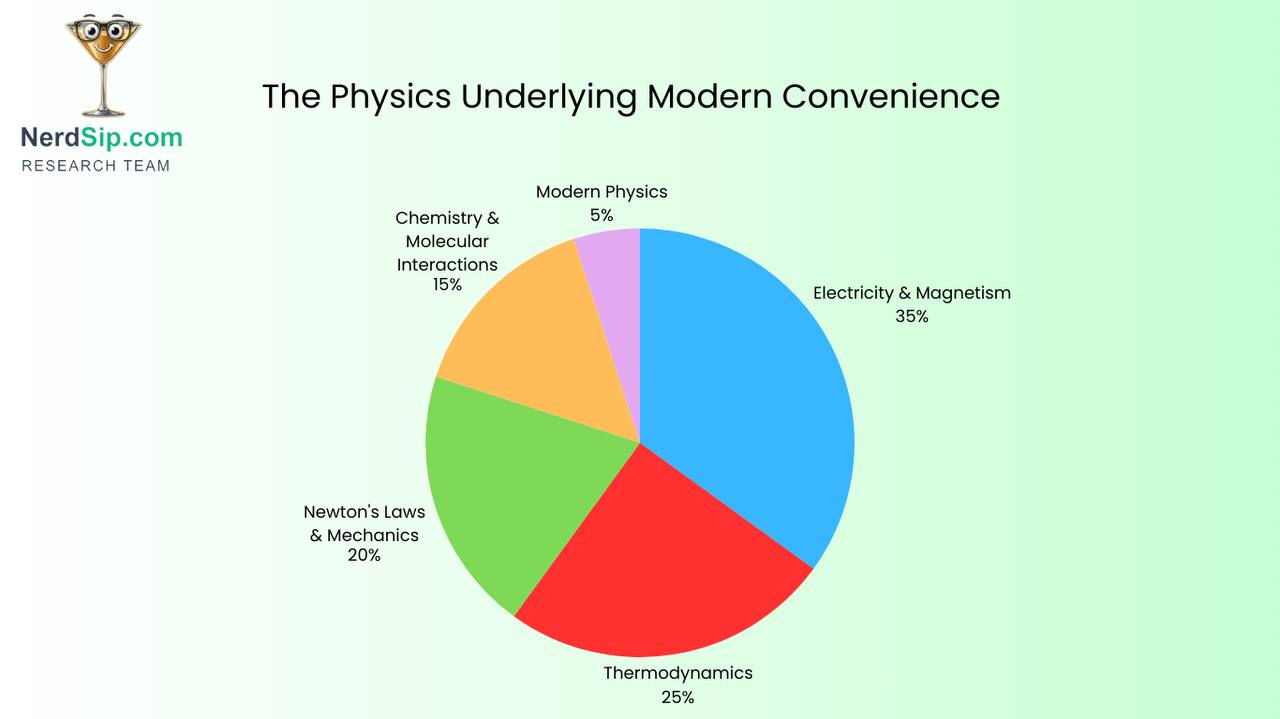
Your microwave is heating your food with the same type of radiation that carries cell phone signals. Not similar to it — the exact same electromagnetic waves, just at a slightly different frequency.
And it's doing this by specifically targeting water molecules. The 2.45 gigahertz frequency it uses happens to be the exact frequency that makes water molecules spin. They rotate billions of times per second, generating friction, which generates heat. That's why a wet paper towel heats up faster than a dry one. Your microwave isn't heating "food" — it's making water molecules go berserk.
Also, the metal grid on your microwave door isn't decorative. The holes are specifically smaller than the wavelength of the microwaves (about 12 centimeters), which means the waves physically cannot pass through. Light has a much shorter wavelength, so it goes straight through and you can watch your food spin. It's a Faraday cage designed to be transparent.
If you just thought "wait, I use this thing every day and had no idea how it works" — you're about to have that feeling about fifteen more times.
Here's how the everyday things around you actually work, and why understanding them makes the world way more interesting.
Why Bread Rises (It's Basically Controlled Fart Gas)
Yeast is alive. That's not metaphorical. When you mix yeast into dough, you're adding millions of single-celled fungi that wake up, eat the sugar in the flour, and excrete carbon dioxide and alcohol as waste products.
The CO₂ gets trapped in the gluten network (the stretchy protein structure formed when you knead dough), creating thousands of tiny bubbles. That's what makes bread fluffy. The alcohol mostly evaporates during baking, though some remains and contributes to the aroma.
So yes: bread rises because you're trapping yeast farts in a protein cage. Baking is just controlled fermentation. Beer and wine use the same process — yeast eating sugar and producing CO₂ and alcohol — except brewers keep the alcohol and let the CO₂ escape. Bakers do the opposite.
Sourdough uses wild yeast and bacteria instead of commercial yeast, which is why it tastes tangy — the bacteria (primarily Lactobacillus) produce lactic acid as a byproduct. That's the same acid that makes yogurt sour and your muscles burn during intense exercise. A healthy sourdough starter contains millions of microorganisms working together in a symbiotic relationship that humans have maintained for over 6,000 years.
Understanding this completely changes how you think about baking. You're not following a recipe. You're managing millions of microscopic organisms to create the texture and flavor you want.
How GPS Knows Exactly Where You Are (Einstein Is Involved)
Your phone is talking to satellites 20,000 kilometres above Earth, comparing the tiny differences in signal arrival time to triangulate your position down to a few metres.
Each GPS satellite contains an atomic clock accurate to within nanoseconds. Your phone receives signals from at least four satellites simultaneously, each one timestamped. Because radio waves travel at the speed of light (exactly 299,792,458 metres per second), your phone can calculate how far away each satellite is based on how long the signal took to arrive.
Three satellites give you latitude and longitude. The fourth corrects for your phone's clock error (your phone's clock isn't atomic-level precise) and gives you altitude.
Here's where it gets weird: the satellites' atomic clocks are adjusted for both special relativity and general relativity. Special relativity says moving clocks run slower — the satellites are moving at about 14,000 km/h, so their clocks lose about 7 microseconds per day. General relativity says clocks run faster in weaker gravity — at 20,000 km altitude, gravity is weaker, so their clocks gain about 45 microseconds per day.
Net effect: satellite clocks run about 38 microseconds fast per day. This is corrected in the system. Without Einstein's relativity corrections, GPS would drift by about 10 kilometres per day and be completely useless within hours.
Your phone casually uses general relativity to tell you where the nearest coffee shop is. Einstein's equations, published in 1915, are running in the background of your navigation app right now.
Why Tyres Are Black (And Why They Grip Better When Warm)
Pure natural rubber is pale, almost white. It's also terrible for tyres — it wears out fast, cracks in sunlight, and gets dangerously slippery when wet.
Tyres are black because they're filled with carbon black, a form of nearly pure elemental carbon made by burning petroleum products in a controlled environment with limited oxygen. It makes up about 30% of a tyre's weight.
Carbon black dramatically improves durability, heat resistance, and grip. It forms a reinforcing structure within the rubber at a molecular level, making the material far stronger. Without it, tyres would last maybe 5,000 kilometres instead of 50,000+. Modern silica compounds are also added to improve wet grip and reduce rolling resistance.
The tread patterns aren't just for show either. They're carefully engineered to channel water away from the contact patch. Without tread, a thin layer of water builds up between tyre and road — hydroplaning — and you lose all grip. Different tread patterns are optimised for different conditions: summer tyres prioritise dry and wet grip, winter tyres have thousands of tiny edges (sipes) to bite into snow and ice, racing slicks have no tread at all because they're only used in dry conditions and maximum contact area equals maximum grip.
Here's why racing tyres need warming up: rubber compounds work through molecular adhesion. Cold rubber is stiff — the polymer chains don't move much. As temperature increases, the chains become more flexible and can conform to microscopic irregularities in the road surface, dramatically increasing grip. Racing tyres are designed to work at 80–100°C. Below that, they're dangerously slippery. This is why race drivers weave before a race — they're generating friction to heat the tyres.
A modern performance tyre is one of the most highly engineered consumer products you interact with regularly, and it's holding your entire car to the road through a contact patch roughly the size of your palm per wheel.
How Aeroplanes Stay Up (And Why the Standard Explanation Is Wrong)
The standard explanation — air moving faster over the curved top of the wing creates lower pressure (Bernoulli's principle), sucking the wing upward — is both partially true and deeply misleading.
The real answer is Newton's third law: the wing deflects air downward, and the air pushes the wing upward with equal force. Aeroplanes fly because they're constantly shoving massive quantities of air toward the ground.
Bernoulli does play a role, but it's not the primary mechanism. The wing shape (airfoil) is designed to redirect airflow downward smoothly. As the wing moves forward, it forces air down. The reaction force is lift. Simple.
This is why planes can fly upside down — aerobatic aircraft are designed with symmetric airfoils and rely purely on angle of attack (the angle between the wing and oncoming air). Tilt the wing up slightly, air gets deflected down, plane goes up. Orientation doesn't matter — physics does.
The misconception persists because Bernoulli sounds more sophisticated and gets taught in schools as the explanation. But ask any aerospace engineer: it's fundamentally about deflecting air mass downward and Newton's laws doing the rest.
Also: commercial jets cruise at around 35,000 feet not because it's "halfway to space" but because air density drops exponentially with altitude. Thinner air means less drag, which means better fuel efficiency. The engines can still get enough oxygen for combustion at that altitude, and the reduced drag more than compensates for the slightly reduced engine efficiency.
Why Phones Get Hot (And Why Wireless Charging Is Worse)
Your phone is a computer. Computers run on electricity. Electricity flowing through circuits encounters resistance. Resistance generates heat.
Every calculation your phone's processor makes involves billions of transistors switching on and off, channeling electrons through microscopic pathways. Not all that electrical energy becomes computation — some becomes waste heat. The faster the processor runs, the more heat it generates.
Modern phone processors are insanely efficient compared to desktop CPUs — often delivering similar performance at 5–10 watts instead of 100+ watts. But they're packed into a space the size of a fingernail with no active cooling (no fans). The heat spreads through the phone's body, which acts as a passive heatsink. The metal or glass back of your phone is literally designed to radiate heat away.
When you're running intensive apps — games, video recording, GPS navigation — the processor works harder and generates more heat. That's normal. Phones are designed to handle this. If they get too hot, they throttle performance to cool down (thermal throttling).
Charging generates additional heat because you're pushing electrical current into a battery, and batteries have internal resistance. Fast charging generates more heat because it's pushing more current. This is why your phone gets warmer during the last 20% of charging if you're using it — it's simultaneously drawing power and trying to charge.
Wireless charging is significantly less efficient — typically 70–80% efficient compared to 90–95% for wired charging. That missing 20–30% becomes heat. The charging pad heats up. Your phone heats up. It's convenient but wasteful, and the extra heat isn't great for battery longevity.
The only time to worry is if your phone becomes painfully hot to hold or starts malfunctioning. That indicates a problem — usually a battery issue or a runaway background process. Normal operation feels warm, not burning.
Why Aeroplane Windows Have That Tiny Hole (It's Not for Equalisation)
Look closely at an aeroplane window. There's a small hole drilled in it, about 1–2 millimetres wide. That's the breather hole, and it's there for pressure management — but not the way most people think.
Aeroplane windows are actually three layers: an outer pane, a middle pane, and an inner scratch pane (the one you touch). The outer pane bears almost all the pressure load — at cruising altitude, the pressure difference between cabin and outside is roughly equivalent to an 8–9 psi difference, or about 3 tonnes of force per square metre.
The breather hole is drilled in the middle pane. It allows air pressure in the gap to equalise with the cabin, which means the middle pane doesn't bear any significant load — it's just a backup. If the outer pane somehow fails (extraordinarily rare), the middle pane can hold cabin pressure temporarily while the pilot descends. The hole also prevents moisture from accumulating and fogging between the panes.
The outer pane has no hole. It's solid acrylic (not glass — acrylic is lighter and more resistant to crack propagation). The small hole you can see from inside is in the middle pane only.
That tiny hole is part of a redundant safety system keeping you pressurised at 10,000 metres altitude while you're watching movies and complaining about legroom.
Why Fizzy Drinks Fizz (And Why Altitude Matters)
Carbon dioxide dissolves in water under pressure. When you open a bottle or can, you release that pressure. The CO₂ becomes less soluble and escapes as gas bubbles. That's the fizz.
The higher the pressure, the more CO₂ stays dissolved. This is Henry's Law: the solubility of a gas in a liquid is proportional to the pressure of that gas above the liquid.
Carbonation happens in factories by chilling the drink (cold liquids dissolve more gas) and forcing CO₂ into it under high pressure — typically 3–4 atmospheres. When sealed, the drink stays fizzy because the pressure keeps the CO₂ dissolved.
Once opened, CO₂ escapes. Shaking accelerates this by creating nucleation sites (tiny bubbles that act as seeds for more CO₂ to escape around). That's why shaken fizzy drinks explode when opened — the liquid is supersaturated with CO₂ looking for an escape route, and the sudden pressure drop gives it one.
Here's something weird: altitude affects carbonation. At higher elevations, atmospheric pressure is lower, which means CO₂ escapes more readily. A drink that's perfectly carbonated at sea level will be more fizzy at altitude. Breweries in places like Denver (1,600m altitude) have to adjust their carbonation levels. Airlines serving fizzy drinks at cruising altitude deal with drinks that are significantly fizzier than they would be on the ground.
Going flat is inevitable after opening. CO₂ will keep escaping until equilibrium with atmospheric CO₂ is reached — which means almost none left in the drink. You can slow it by recapping tightly (reducing air contact) and refrigerating (cold liquids hold CO₂ better), but you can't stop it.
The same principle explains why champagne gets you drunk faster: the CO₂ bubbles speed up alcohol absorption in your stomach by increasing the surface area exposed to the stomach lining and slightly irritating it, increasing blood flow. It's not a myth — carbonation measurably increases the rate at which alcohol enters your bloodstream.
How Bluetooth Got Its Name (And What the Logo Actually Means)
Bluetooth — the short-range wireless technology in your headphones, speakers, and car — is named after Harald "Bluetooth" Gormsson, a 10th-century Viking king who united Denmark and Norway.
The technology was developed in the 1990s by a Swedish company (Ericsson) and needed a codename during development. An engineer named Jim Kardach suggested "Bluetooth" as a placeholder, referencing the king who united Scandinavia, because the technology was meant to unite different communication protocols and allow devices to talk to each other.
The name was only supposed to be temporary. They planned to call it something else at launch. But by the time they needed to pick a final name, "Bluetooth" had stuck internally and they couldn't find anything better. So a temporary codename based on a Viking king became one of the most widely deployed wireless standards in history.
The Bluetooth logo is literally a combination of the runes for Harald's initials: ᚅ (Hagall, H) and ᚢ (Berkanan, B), merged together into a single symbol. Look at the logo now. You can't unsee it.
Why was the king called Bluetooth? Most likely because he had a conspicuously dead or discoloured tooth that looked dark blue or grey. Medieval dental care was not great. So you're connecting your phone to your car using a protocol named after a Viking king's rotten tooth.
History is weird, and the person who named Bluetooth had a sense of humor.
Why Microwaves Don't Work on Metal (But Sometimes Do)
You've been told never to put metal in microwaves. That's mostly correct but not entirely accurate.
Microwaves are electromagnetic radiation. Metals are full of free electrons that respond immediately to electromagnetic fields. When microwaves hit metal, the electrons oscillate rapidly, creating currents and heat. If the metal has sharp points or edges, the currents can build up and create sparks (arcing) — which damages the microwave and potentially starts a fire.
Flat metal can actually be fine. Many microwave ovens have metal racks designed for use inside them. The key is that flat metal without sharp edges reflects microwaves but doesn't concentrate currents enough to create arcing.
Aluminum foil, in small amounts and used carefully (no crinkled edges), won't hurt anything. Professional chefs sometimes use foil to shield parts of food from overcooking. But crinkled foil with sharp points will definitely spark.
The rotating plate in your microwave exists because microwaves create standing wave patterns inside the oven — there are hot spots and cold spots. Rotation ensures food moves through all zones and heats more evenly. Some microwaves use a rotating antenna instead of a rotating plate to distribute the waves more uniformly.
Also: microwaves don't cook from the inside out. That's a complete myth. They penetrate food to a depth of roughly 1–2 centimeters, depending on water content. The inside of thick food is cooked by heat conducting inward from the outer layers, same as conventional cooking. This is why microwave instructions often tell you to let food stand after heating — the heat is still conducting inward.
Why Fridges Are Cold Inside and Hot Behind
Your refrigerator is not creating cold. It's removing heat. Big difference.
A fridge uses a refrigerant (a special fluid designed to evaporate and condense at low temperatures) and a compressor to move heat from inside the fridge to outside. Here's the cycle:
- Inside the fridge: Liquid refrigerant passes through an expansion valve, suddenly dropping pressure. This causes it to evaporate rapidly, and evaporation absorbs heat (same reason sweat cools you down). The evaporating refrigerant absorbs heat from inside the fridge, making it cold.
- The refrigerant (now a gas) flows to the compressor at the back of the fridge. The compressor squeezes it, dramatically increasing pressure and temperature.
- The hot, pressurized gas flows through coils on the back of the fridge (or underneath), where it releases heat to the room and condenses back into liquid.
- The liquid refrigerant returns to the expansion valve and the cycle repeats.
This is why the back of your fridge is warm — it's radiating the heat removed from inside. A fridge doesn't make cold; it moves heat from inside to outside.
This is also why you can't cool a room by leaving the fridge door open. The fridge will move heat from inside the fridge to the back coils, but all that heat (plus the waste heat from the compressor motor) goes into the room. Net effect: the room gets warmer.
Air conditioners work exactly the same way — they're just fridges with the inside coil in your house and the outside coil... outside.
Where Curious People Go From Here
These aren't party tricks. Each one is a doorway into deeper understanding.
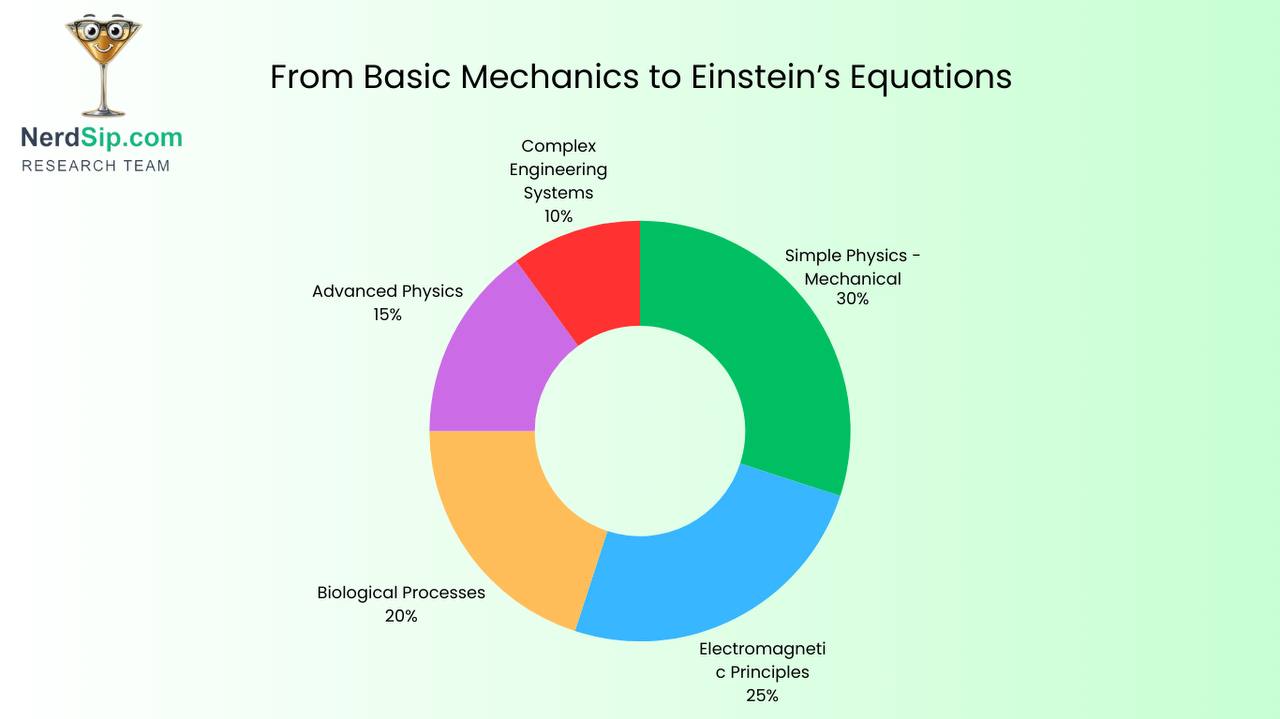
Microwaves lead to electromagnetic radiation broadly — how WiFi works, why some materials conduct electricity and others don't, how radio telescopes detect distant galaxies.
Yeast and fermentation lead to biochemistry and cellular respiration — how your own cells extract energy from food, why anaerobic exercise feels different, how antibiotics target bacteria without harming your cells.
GPS leads to relativity, atomic clocks, and orbital mechanics — the physics governing everything from satellite internet to interplanetary missions to why astronauts age slightly slower in orbit.
Tyres lead to polymer chemistry and tribology (the study of friction) — why different materials stick or slide, how joints and bearings work, why some surfaces feel smooth and others rough.
Aeroplanes lead to fluid dynamics — how heart valves work, why hurricanes are low-pressure systems, how wings evolved in birds and insects independently.
Phone heat leads to semiconductor physics and Moore's Law — how transistors work, why chips can't get infinitely faster, what quantum computing is trying to solve.
Every mundane object around you is the end result of centuries of accumulated engineering and science. Understanding how they work doesn't make them less magical — it makes them more.
Apps like NerdSip are built for this exact kind of curiosity. Want to understand how your phone actually works? How fermentation creates alcohol and bread? How relativity affects your daily life? Generate a course in seconds. Five to ten minute lessons. Follow what interests you.
Because the world is running on principles you can understand. You just have to look.
The science is everywhere. Go find it.
📚 Keep Learning
Ready to Level Up?
Turn your screen time into knowledge. Download NerdSip and start learning in just 5 minutes a day.



